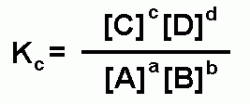Equilibrium
In stoichiometry calculations, we assume that reactions run to completion. However, when a chemical reaction is carried out in a closed vessel, the system achievesequilibrium. Equilibrium occurs when there is a constant ratio between the concentration of the reactants and the products. Different reactions have different equilibria. Some may appear to be completely products, however, all reactions have some reactants present. A reaction may look "finished" when equilibrium is reached, but actually the forward and reverse reactions continue to happen at the same rate. A reverse reaction is when the written reaction goes from right to left instead of the forward reaction which proceeds from left to right. This is why equilibrium is also referred to as "steady state".
Le Chatelier's Principle
Le Chatelier's principle states that when a system in chemical equilibrium is disturbed by a change of temperature, pressure, or a concentration, the system shifts in equilibrium composition in a way that tends to counteract this change of variable. The three ways that Le Chatelier's principle says you can affect the outcome of the equilibrium are as follows:
- Changing concentrations by adding or removing products or reactants to the reaction vessel.
- Changing partial pressure of gaseous reactants and products.
- Changing the temperature.
Equilibrium Constant
To determine the amount of each compound that will be present at equilibrium you must know the equilibrium constant. To determine the equilibrium constant you must consider the generic equation:
aA + bB <--> cC + dD
The upper case letters are the molar concentrations of the reactants and products. The lower case letters are the coefficients that balance the equation. Use the following equation to determine the equilibrium constant (Kc).
aA + bB <--> cC + dD
The upper case letters are the molar concentrations of the reactants and products. The lower case letters are the coefficients that balance the equation. Use the following equation to determine the equilibrium constant (Kc).
Course Notes on Power Point
| chemical_equilibrium.ppt | |
| File Size: | 3021 kb |
| File Type: | ppt |
| le_chatelier_principle.ppt | |
| File Size: | 3222 kb |
| File Type: | ppt |
| developing_a_problem_solving_strategy_for_chemical_equilibrium_problems.docx | |
| File Size: | 19 kb |
| File Type: | docx |
Ksp of calcium hydroxide lab |
| ||||||||||||
| solubility_equilibrium_summary.docx | |
| File Size: | 16 kb |
| File Type: | docx |
|
| ||||||||||||||||||
| acid-base_titration.ppt | |
| File Size: | 100 kb |
| File Type: | ppt |
| acid_base_summary.docx | |
| File Size: | 24 kb |
| File Type: | docx |